First Multiple Sclerosis Pill approved by FDA
Posted on September 28, 2010 by Jeffrey Newman
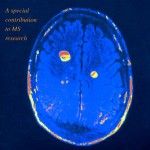 Last week, Novartis received FDA approval to market the once a day pill Gilenya, a sphingosine 1-phosphate receptor modulator for treatment of multiple sclerosis. Other MS treatments require injection. The precise mechanism of action is not understood in MS but seems to act by reducing the release of white cells from lymph nodes thereby reducing the white blood cell influx to the central nervous system. Gilenya has been tested in over 2,600 patients and demonstrates reduction in relapse rate and active lesions as measured by MRI compared to placebo in a two year trial. It is a once a day pill. MS is an autoimmune disease in which a person’s immune system attacks the myelin sheaths covering the neuronal axons in the central nervous system. It is extracted from a fungus and has some side effects.
Last week, Novartis received FDA approval to market the once a day pill Gilenya, a sphingosine 1-phosphate receptor modulator for treatment of multiple sclerosis. Other MS treatments require injection. The precise mechanism of action is not understood in MS but seems to act by reducing the release of white cells from lymph nodes thereby reducing the white blood cell influx to the central nervous system. Gilenya has been tested in over 2,600 patients and demonstrates reduction in relapse rate and active lesions as measured by MRI compared to placebo in a two year trial. It is a once a day pill. MS is an autoimmune disease in which a person’s immune system attacks the myelin sheaths covering the neuronal axons in the central nervous system. It is extracted from a fungus and has some side effects.
