DRUG TRIALS TO EXPLORE SLOWING ALZHEIMER’S
Posted on July 18, 2010 by Jeffrey Newman
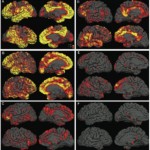 New drugs trials are being set up to try to halt the development of amyloid plaque accumulations at the earliest stages of Alzheimer’s disease, before significant brain damage occurs. An issue with the trials has been raised by the FDA which says it must know not just that plaque was reduced or eliminated but also that those who took the drug ended up with better memories and abiliities to think and reason compared to those who did not take the drug. A Harvard Researcher, Dr. Dennis Selkoe says that the study, underwitten bu Bristol-Myers, will include examination on syptoms like memory and reasoning at the ame time tracking amyloid in the brain. It is somewhat of a risk because even people with fairly mild symptoms may already have too much brain damage to be helped. Alzheimer’s disase afflicts more than 5.3 million Americans and is the seventh leading cause of death. Amyloid beta is a rare gene mutation which leads t overproduction. Downs syndrome also causes overproduction of amyloid beta and people with Down always get Alzheimer’s. Scientists are not at the point yet where they are sure that amyloid beta causes the disease. Dr, Paul Aiden of the University of California in San Diego is preparing a large federal study to test amyloid-blocking drugs in people over 70 with normal memories but some evidence of amyloid accumulation. He will focus on whether the drugs slow brain atrophy which often happens in Alzheimer’s. It is expected to take ten years to know the key results. sources: NY Times; Science .
New drugs trials are being set up to try to halt the development of amyloid plaque accumulations at the earliest stages of Alzheimer’s disease, before significant brain damage occurs. An issue with the trials has been raised by the FDA which says it must know not just that plaque was reduced or eliminated but also that those who took the drug ended up with better memories and abiliities to think and reason compared to those who did not take the drug. A Harvard Researcher, Dr. Dennis Selkoe says that the study, underwitten bu Bristol-Myers, will include examination on syptoms like memory and reasoning at the ame time tracking amyloid in the brain. It is somewhat of a risk because even people with fairly mild symptoms may already have too much brain damage to be helped. Alzheimer’s disase afflicts more than 5.3 million Americans and is the seventh leading cause of death. Amyloid beta is a rare gene mutation which leads t overproduction. Downs syndrome also causes overproduction of amyloid beta and people with Down always get Alzheimer’s. Scientists are not at the point yet where they are sure that amyloid beta causes the disease. Dr, Paul Aiden of the University of California in San Diego is preparing a large federal study to test amyloid-blocking drugs in people over 70 with normal memories but some evidence of amyloid accumulation. He will focus on whether the drugs slow brain atrophy which often happens in Alzheimer’s. It is expected to take ten years to know the key results. sources: NY Times; Science .
